Tube-Plate Sealing Strategies
Candidate: Tianyu Bai(Dartmouth)
Required Hardware Components
Empirical Prototyping & Scaled Feasibility
Macro-scale physical simulations and 1:1 rapid prototyping to physically validate spatial constraints and identify macroscopic failure modes.




Strategic Mechanism Analysis
Feasibility evaluation of sealing modalities relative to the 1 mm spatial limit and 0.1 mm eccentricity.
First-Principles Physics & Fluid Dynamics
Mathematical validation of capillary imbibition, microbubble genesis, and thermo-mechanical limits.
1. Capillary Driving Force (Laplace)
Spontaneous liquid penetration driven by the pressure differential across the curved meniscus.
2. Imbibition Time (Washburn)
Filling time is proportional to fluid viscosity and the square of the target depth (\(L^2\)).
3. Microbubble Trapping (Stokes)
Terminal ascending velocity plunges proportionally to the square of a bubble's shrinking diameter.
4. Thermo-Mechanical Shear
Internal stress generated by the CTE mismatch between 316SS and the epoxy matrix during heating.
Sealing Material: Master Bond EP42HT-2Med
The injection process utilizes EP42HT-2Med, a medical-grade epoxy known for high-temperature resistance and superior biocompatibility.
Interactive Operation Simulation
Injection Process Overview
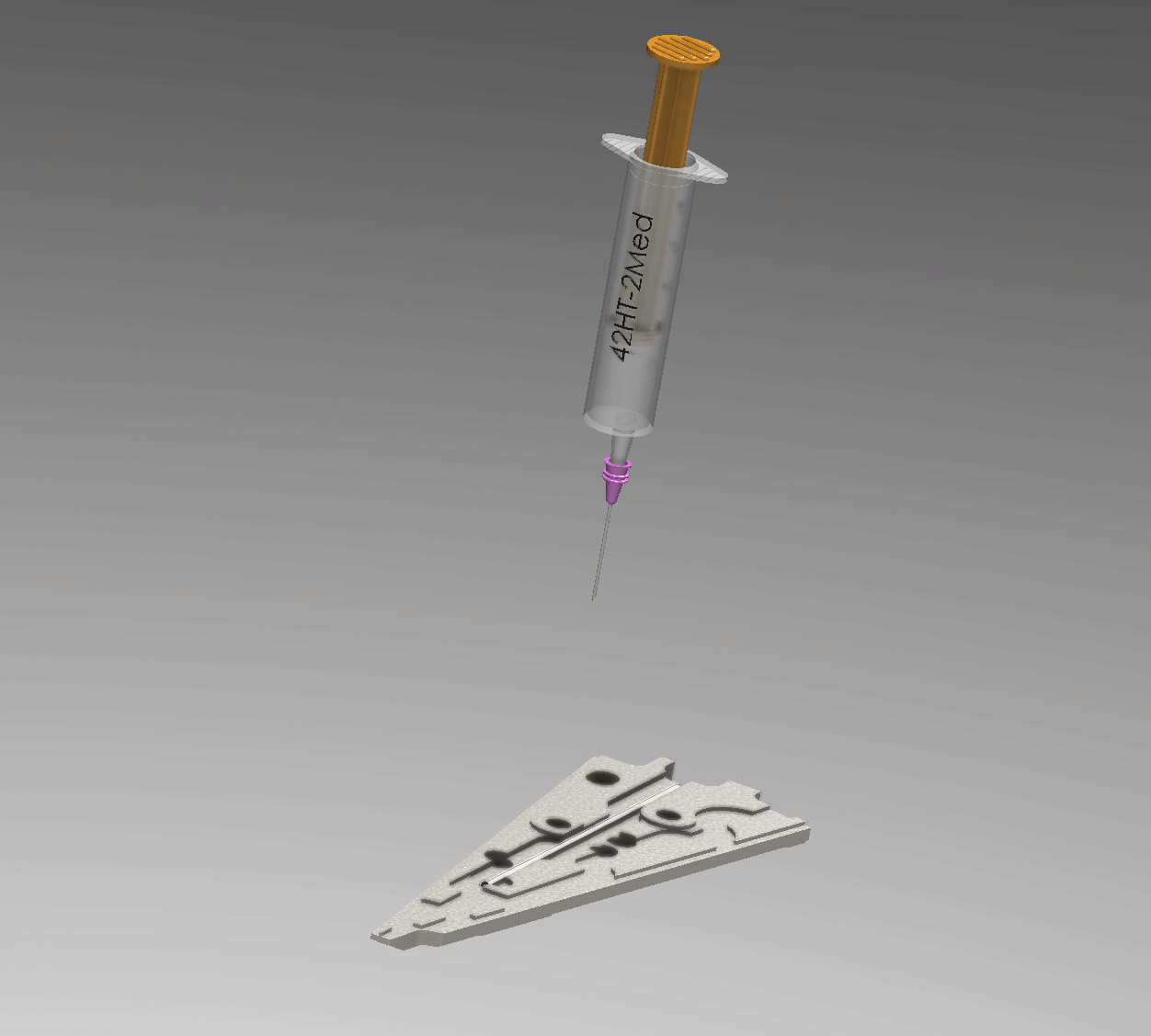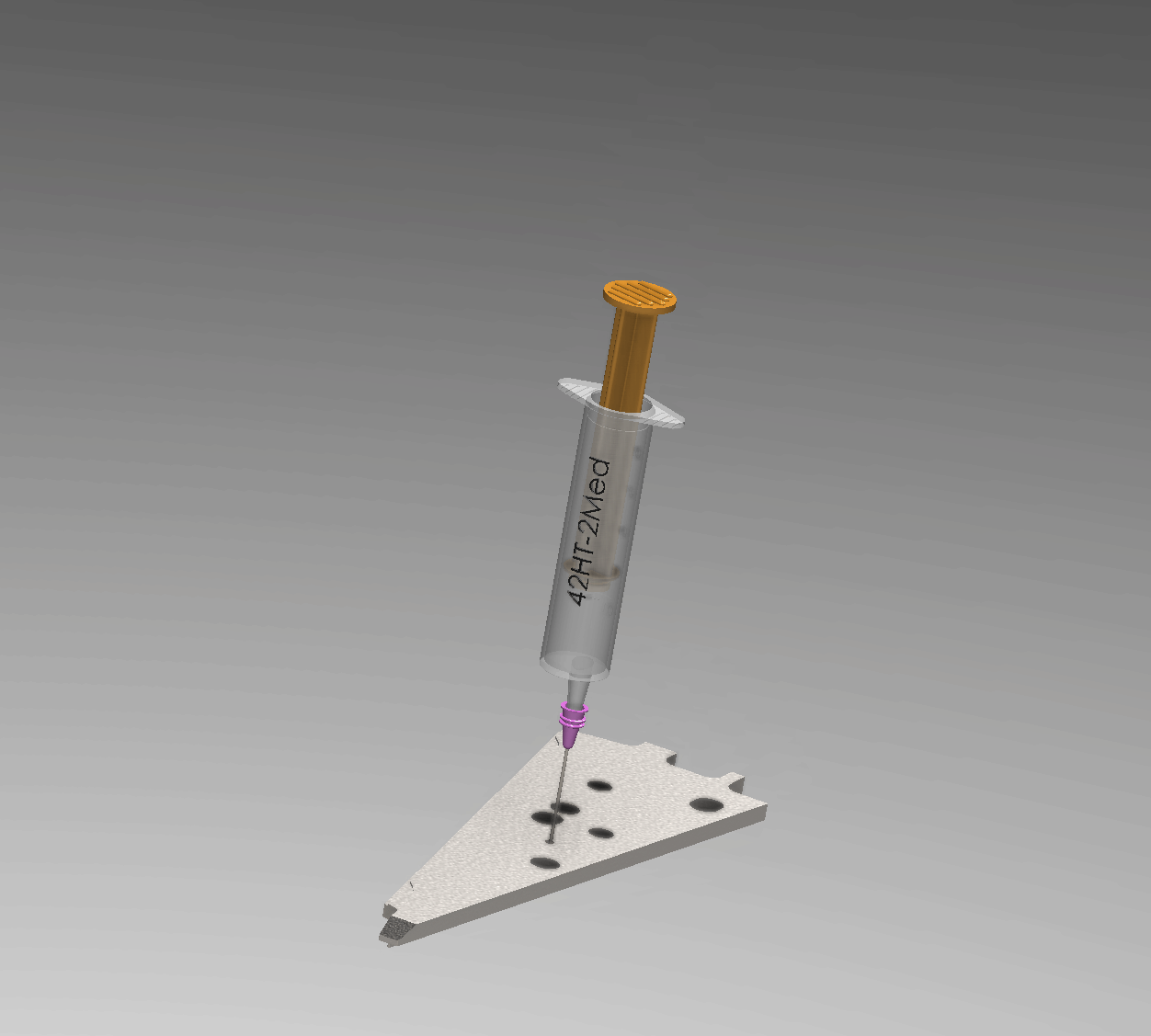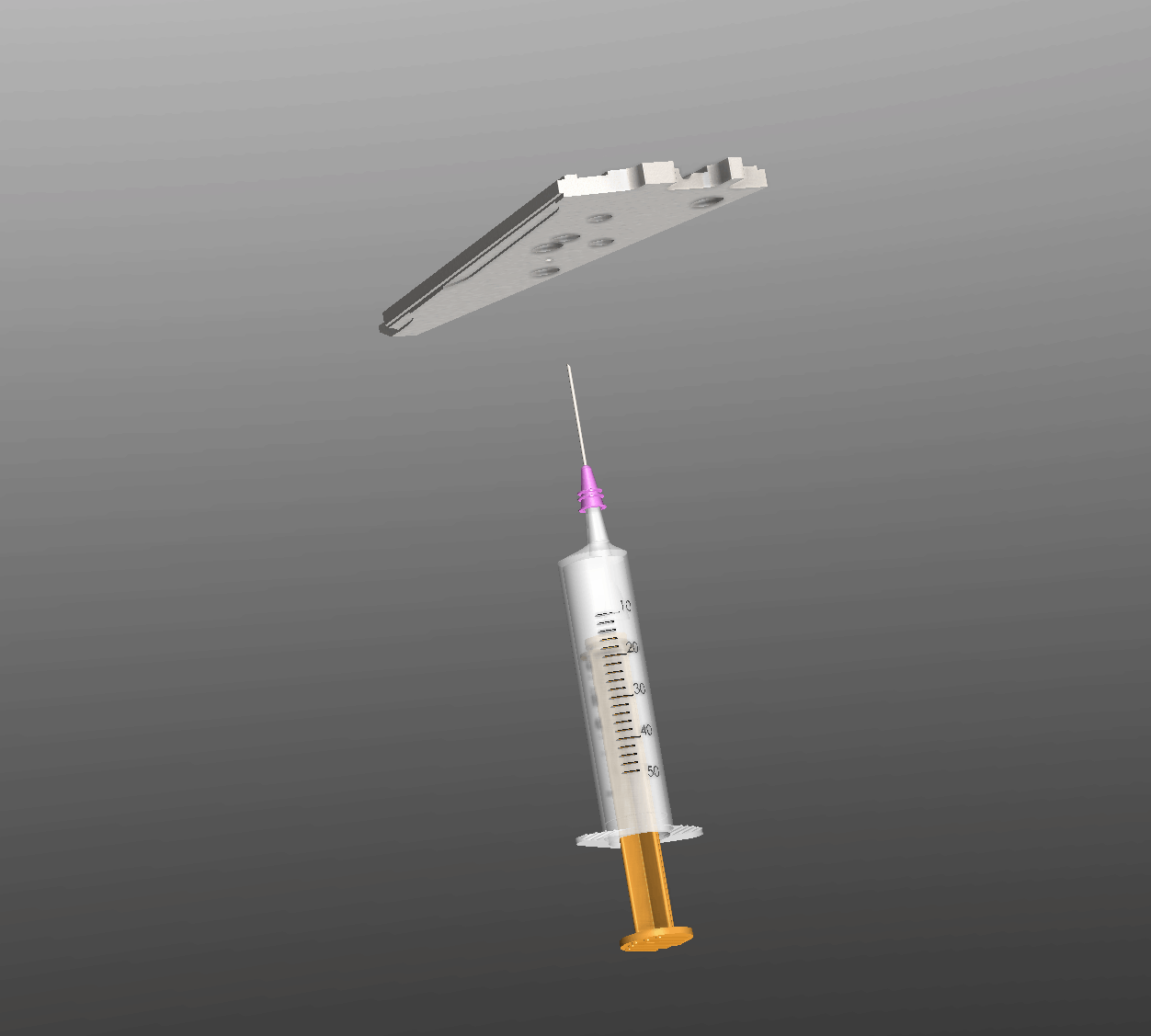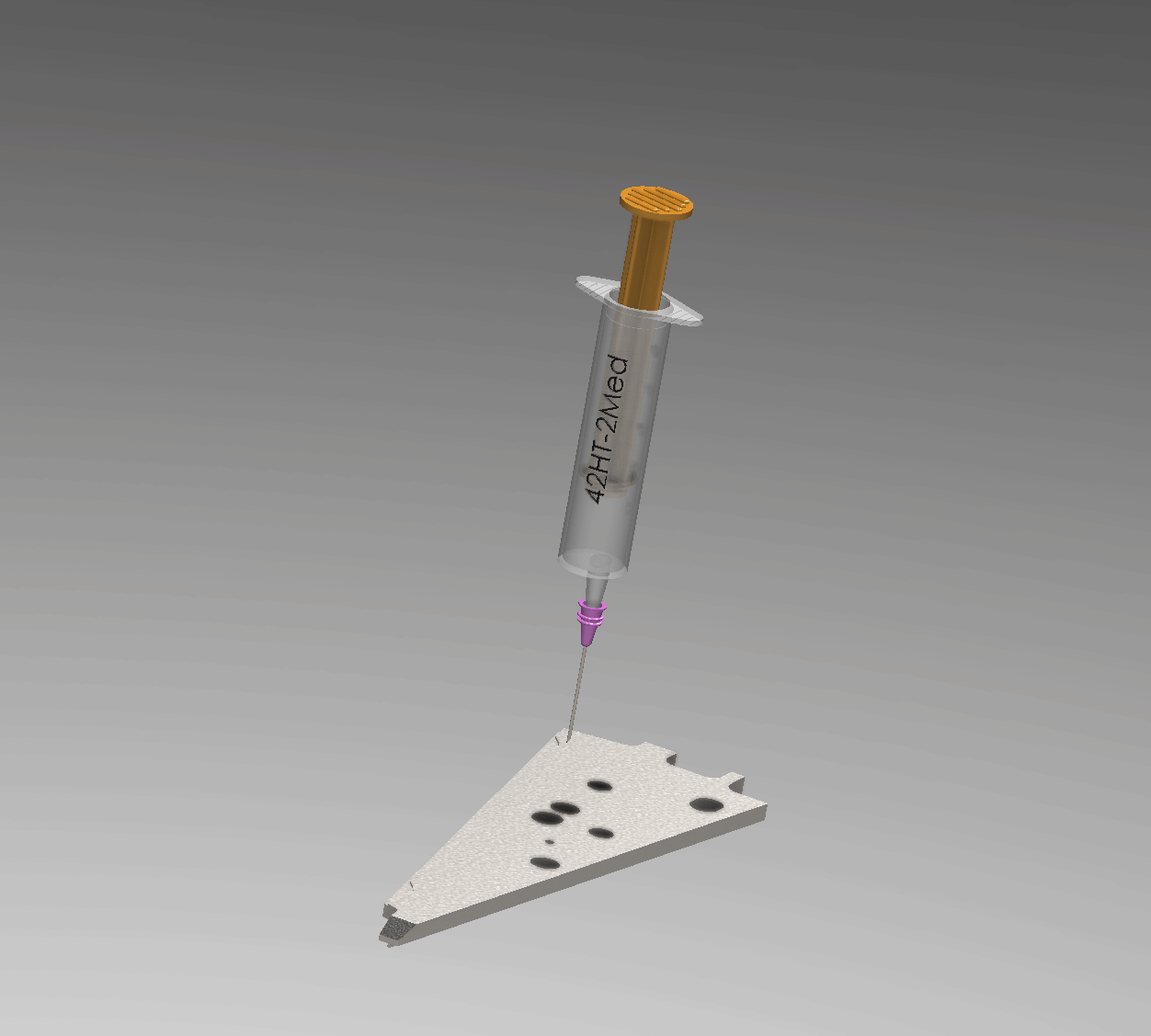
Manufacturing Engineering Workflow
A precise 6-step process ensures a void-free hermetic seal within the 0.1 mm annular gap.
Activation
Degassing
Tuning
Dispensing
Curing
Test
Structural Integrity & Safety Margins
Validation based on First-Principles of solid mechanics and ASTM standards.
THERMO-MECHANICAL COMPATIBILITY VERIFICATION
Result: Confined radial expansion neutralizes shear strain. The ΔCTE instead induces an 840 psi compressive normal stress, synergistically enhancing the hermetic seal at 121°C without exceeding yield limits.
Quality Validation & Standards
Strict adherence to international standards for neurosurgical sterile integrity.